Abstract
Background
The development of mRNA sequencing has contributed greatly to the mechanism exploration in hematologic malignancies disease. With the advent of revolutionized single-cell mRNA sequencing (scRNA-seq), it is now possible to characterize every subset of expression programs and functional states in a comprehensive and unbiased manner. Here, we present a systematic evaluation of engineered chimeric antigen receptor T (CAR-T) products and patient bone marrow profiles in terms of primary resistance and severe cytokine release syndrome (CRS) at the single-cell level.
Methods
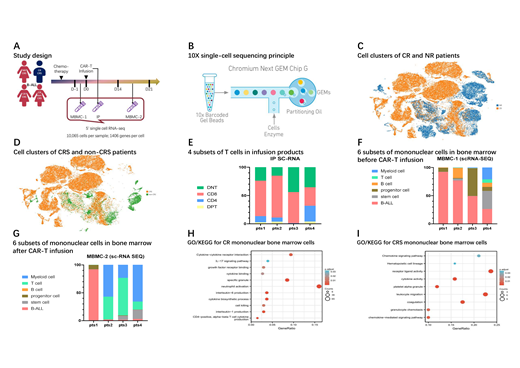
Using single-cell mRNA sequencing in conjunction with flow cytometry (FCM), we performed characterization of CD19-targeted CAR-T and mononuclear bone marrow cells from 4 on-trial B acute lymphoblastic leukemia (B-ALL) patients (NCT02965092). Bioinformatics analysis was utilized to explore diversity between patients with different grades of response or CRS. Basing on marker genes, CAR-T products were divided into four groups, which were double-positive T (DPT), CD4 positive T (CD4), CD8 positive T (CD8), and double-negative T (DNT) cells. Meanwhile, both the mononuclear bone marrow cells before and after CAR-T infusion were grouped into six clusters, which were B-ALL, stem, progenitor, B, T, and myeloid cells. The expression and enrichment analyses results were calculated by R (version 3.6.3) and then verified in a 22-sample conventional transcription sequencing cohort of the same clinical trial. Patient efficacy was assessed by the national comprehension cancer network guidelines version 2.2020 for acute lymphoblastic leukemia, and CRS was graded by CTCAE 5.0.
Results
By FCM detection, the variances of CAR-T infusion products between patients with different clinical outcomes were limited, and nor did mononuclear bone marrow cells. The scRNA sequencing results showed that distinct CAR-T and bone marrow cell subsets indicated differentiated expression in proliferation, cytotoxicity, and intercellular signaling pathways. Expression differentiation variances in CAR-T infusion products were minor than in mononuclear bone marrow cells. CD8+ CAR-T products of complete response (CR) patients were still significantly enriched in pathways such as cell killing (p adjust=0.0012), antigen processing and presentation (p adjust=0.0027), and cell cycle (p adjust=0.0231), exhibiting greater immune function when compared with no response patients. Also, DPT CAR-T products of the non-CRS patients were meaningfully enriched in negative regulation of cytokine production pathway (p adjust=0.0127) when compared with CRS ones. In mononuclear bone marrow cells, B-ALL cells before CAR-T treatment of CR patients presented negatively in cell-cycle (p adjust=0.0019), leading to a low malignant cell proliferation level; and stem-progenitor cells after CAR-T treatment of CR patients showed a stronger ability of neutrophil activation (p adjust<0.0001). As with comparisons between CRS and non-CRS, B-ALL cells before infusion manifested a cell cycle arrest profile (p adjust<0.0006) in non-CRS patients, whereas the immune cells at the same time point were enriched in positive regulation of cell cycle process (p adjust=0.0002).
Conclusions
Through single-cell RNA-seq profiling and unbiased canonical pathway analyses, our results unveil heterogeneities in the cell cycle, immune phenotype, and metabolic profiles of subsets during CAR-T therapy, providing a mechanistic basis for ameliorating clinical outcomes and individualized management.
No relevant conflicts of interest to declare.